BIOMOLECULES
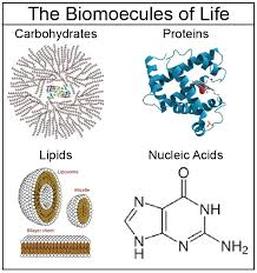
Biomolecules is the molecules of life. Four main classes of biomolecules: carbohydrates, lipids, proteins and nucleic acids.
Carbohydrates, proteins and lipids are huge and therefore called macromolecules.
Carbohydrates, proteins and lipids are huge and therefore called macromolecules.
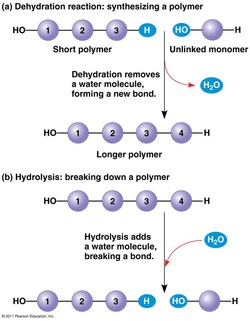
- Macromolecules are polymers, built from monomers.
- A polymer is a long molecule consist of many repeating units of monomers as their building blocks.
- A monomer is a small molecule.
1. Carbohydrates
 Carbohydrates
Carbohydrates
General Structure
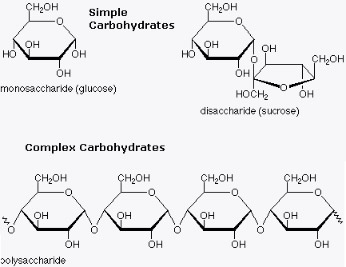
Also known as sugar, carbohydrates serve as fuel and building blocks. Carbohydrates divided into two types that is simple (monosaccharides & disaccharides) and complex sugar (polysaccharides). Molecular formula for monosaccharides are some multiple unit of CH2O. Such as glucose, C6H12O6. The general formula of disaccharides is Cn(H2O)n-1 and trisaccharides is Cn(H2O)n-2 and so on.
- Monosaccharides: glucose, fructose and galactose.
- Disaccharides: maltose, lactose and sucrose.
- Polysaccharides: cellulose, starch, fibers and glycogen.
Linkages
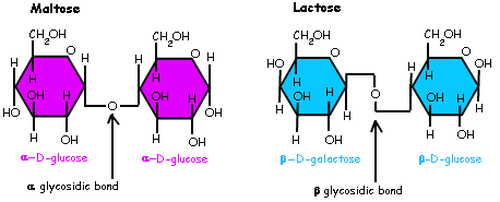
Disaccharides consists of two monosaccharides joined by glycosidic linkage. While polysaccharides are macromolecules with a few thousand monosaccharides also joined by glycosidic linkage.
- Glycosidic linkages is a covalent bond formed between two monosaccharides by a dehydration reaction.
- For example maltose is a disaccharide formed by linking of two molecules of glucose. While lactose is formed by linking of galactose and glucose.
- Function of polysaccharides are determined by its sugar monomers and by the positions of its glycosidic linkages.
Functional group
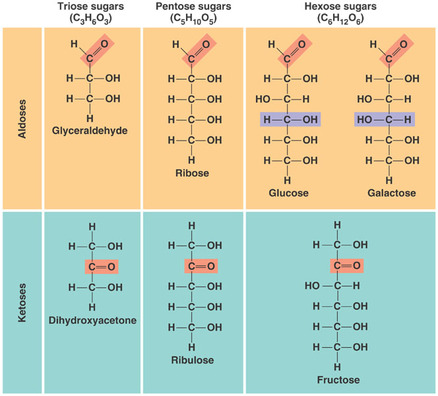
In glucose, the molecules has a carbonyl group (C=O) and multiple hydroxyl group (-OH).
In glucose, the molecules has a carbonyl group (C=O) and multiple hydroxyl group (-OH).
- The types of sugar depends on the location of the carbonyl group either its an aldose (aldehyde sugar) or a ketose (ketone sugar).
- Sugars can be classified based on their size of the carbon skeleton, which trioses (three-carbon), pentoses (five-carbon) and hexoses (six-carbon).
- Spatial arrangement of their parts around asymmetric carbons also give rise to different types of sugar such as glucose, galactose and fructose that have different arrangement of carbonyl and hydroxyl group.
2. Lipids
There are three importants types of lipids: fats, phospholipids and steroid. Lipids not consider as a macromolecules since its not include true polymers.
General Structure
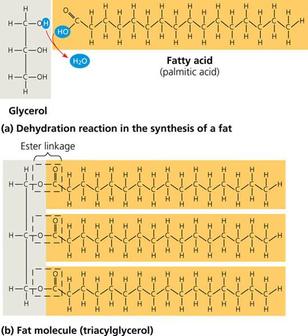
Fats
General Structure
Fats
- Major function as energy storage and protect the internal organ of organism.
- Constructed from dehydration of glycerol (alcohol) and fatty acids. Triacylglycerol is the combination of one glycerol and three fatty acids by ester linkage.
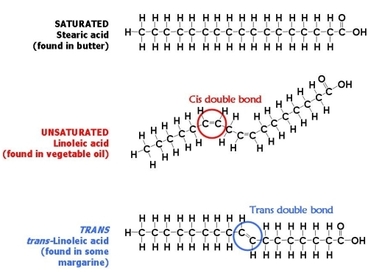
- Fats can be classified as saturated and unsaturated fat.
- Saturated fat have no double bond and have as many hydrogen atoms bonded to carbon skeleton.
- Unsaturated fat have double bonds and fewer hydrogen atoms which cause kink in hydrocarbon chain.
- Trans fat is unsaturated fat with trans double bond that behave like saturated fat. Over consumed will lead to heart disease problem.
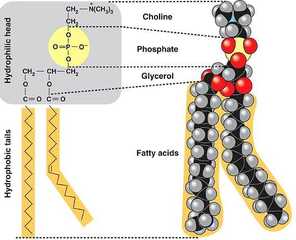
Phospholipids
- Most important type for cell existence as a cell membranes. Arranged into double-layer called "bilayer".
- Constructed from one glycerol and two fatty acids. Phosphate group attached on third hydroxyl group of glycerol.
- Choline group attached to phospholipids (phosphatidylcholine).
- Hydrophobic tails: hate water
- Hydrophilic head: love water
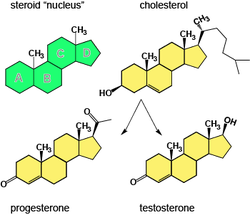
Steroids
- Have four rings of carbon atom. The types of steroids (cholesterol and vertebrate sex hormone) distinguished by chemical groups attached to the rings.
- Cholesterol (component of cell membrane) is the precursor for other steroids.
- Human sex hormone are steroids, a type of hydrophobic compound.
- High level of cholesterol in the blood will lead to atherosclerosis (heart problem).
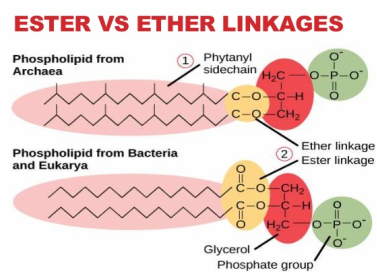
Linkages
- There are two types of linkage in lipids: ester and ether linkage.
- Ester linkage: a bond between hydroxyl and carboxyl group.
- Ether linkage: make archae survive in extreme environment (acidic, alkaline, hot)
- Major functional group for lipids are ester that consists of hydroxyl and carboxyl group.
- Differ in phospholipids, there is phosphate group (-ve charge) attached on the third hydroxyl group of glycerol.
3. Proteins
Proteins have variety of roles some are function as catalyze chemical reaction, structural support, store amino acids, transport substance, defense, movement or cellular communication.
General Structure
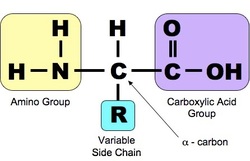
Amino acids are the main/only molecules that can be diverse to become a protein. Protein constructed from the same set of 20 amino acids and consists of one or more polypeptides, each folded and coiled into specific three-dimensional structure. Amino acid is an organic molecule and there are asymmetric carbon atom (alpha carbon) that consists of four different group attached to its. Those functional groups are an amino acids, a carboxylic acid, a hydrogen atom and a variable group R (side chain).
General Structure
Amino acids are the main/only molecules that can be diverse to become a protein. Protein constructed from the same set of 20 amino acids and consists of one or more polypeptides, each folded and coiled into specific three-dimensional structure. Amino acid is an organic molecule and there are asymmetric carbon atom (alpha carbon) that consists of four different group attached to its. Those functional groups are an amino acids, a carboxylic acid, a hydrogen atom and a variable group R (side chain).
Linkages
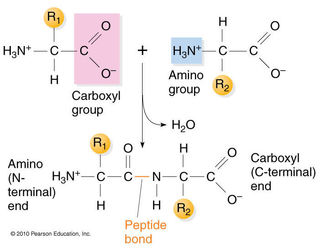
The combination of one or more amino acids (monomers) by dehydration reaction (remove water) between the amino group (H-N-H) and carboxyl group (O=C-OH) will yields a polypeptides chain (polymers). The linkage that formed between the amino group and carboxyl group called as peptide linkage/peptide bond (covalent bond).
The combination of one or more amino acids (monomers) by dehydration reaction (remove water) between the amino group (H-N-H) and carboxyl group (O=C-OH) will yields a polypeptides chain (polymers). The linkage that formed between the amino group and carboxyl group called as peptide linkage/peptide bond (covalent bond).
4. Nucleic Acids
There are two types of nucleic acids, deoxyribonucleic acid (DNA) and ribonucleic acid (RNA). Nucleic function as hereditary information storage (DNA), various function during gene expression (RNA). From DNA transcription and translation, RNA was synthesized and then trigger the formation of protein.
DNA --> RNA --> protein
DNA --> RNA --> protein
General Structure
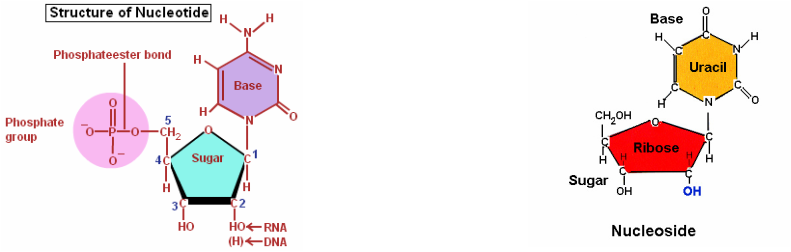
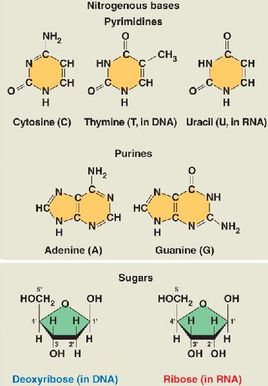
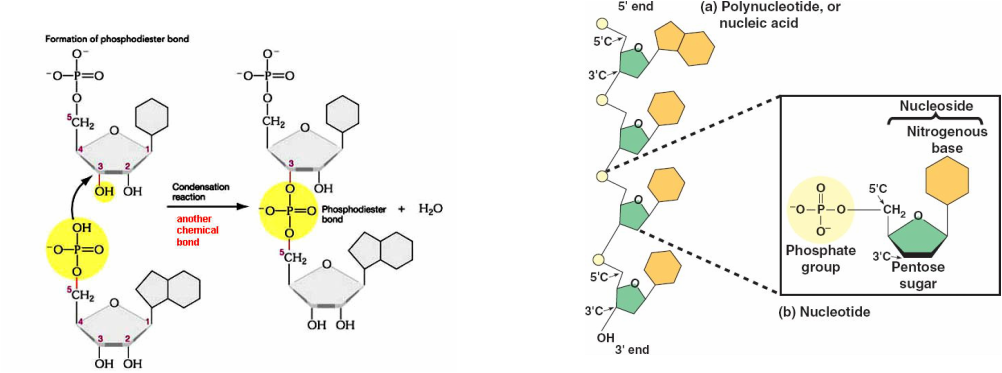
Nucleic acids are macromolecules that formed from the combination of one or more monomers called as polynucleotides. The monomers of nucleic acids are known as nucleotides, consists of three functional groups: a nitrogenous base, a five-carbon sugar (pentose) and one or more phosphate group. Nucleotides without phosphate group are called as nucleoside.
Nucleic acids are macromolecules that formed from the combination of one or more monomers called as polynucleotides. The monomers of nucleic acids are known as nucleotides, consists of three functional groups: a nitrogenous base, a five-carbon sugar (pentose) and one or more phosphate group. Nucleotides without phosphate group are called as nucleoside.
Nucleoside components are nitrogenous base and sugar. Nitrogenous base either one or two rings group as pyrimidines (one ring) and purines (two rings):
- Pyrimidines (six-membered ring): Cytosine (C), Thymine (T, in DNA), and Uracil (U, in RNA).
- Purines (six-membered + five-membered ring): Adenine (A) and Guanine (G).
- Deoxyribose (DNA)
- Ribose (RNA)
Linkages
The combination of one or more nucleotides between pentose sugar (-OH group) with phosphate group by dehydration reaction (remove water) forming phosphodiester linkage/bond. The chain that formed called as polynucleotides (nucleic acids).
The combination of one or more nucleotides between pentose sugar (-OH group) with phosphate group by dehydration reaction (remove water) forming phosphodiester linkage/bond. The chain that formed called as polynucleotides (nucleic acids).