|
|
|
|
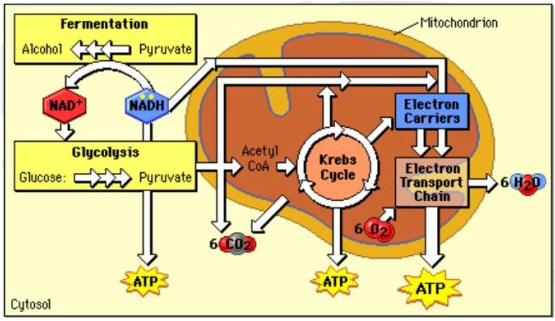
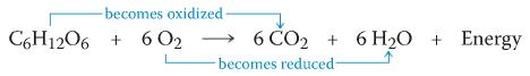
Oxidation-reduction reaction are also known as redox reaction that occurs in cellular respiration and fermentation. Redox reaction is the main chemical reaction that involves the transfer of electron from one reactant to another. The relocation of electrons releases energy, then used to synthesize ATP. Adenosine triphosphate (ATP) works in many ways in cellular respiration. While the Law of Thermodynamics is an organism's chemical reactions called as metabolism. These three properties are related to each other in biological process.
|
Oxidation-reduction (redox reaction)
Redox reaction have two parts:
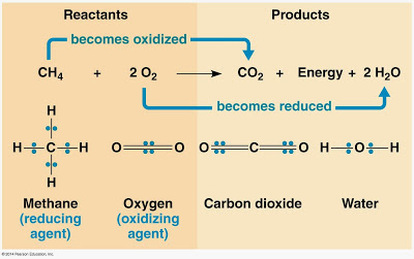
- Oxidation: loss of electrons; substance that donor electron called as reducing agent.
- Reduction: addition of electrons; substance that accept electron called as oxidizing agent.
For example, methane react with oxygen to yield energy by combustion.
Roles of Adenosine Triphosphate (ATP)
|
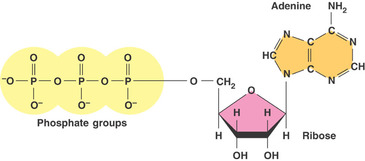
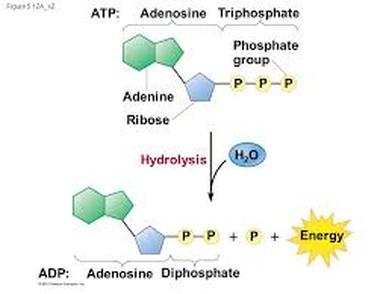
Can be described as energy of life, ATP is the example of nucleic acid as they have three functional group: three phosphate group, sugar ribose and adenine (nitrogenous base).
|
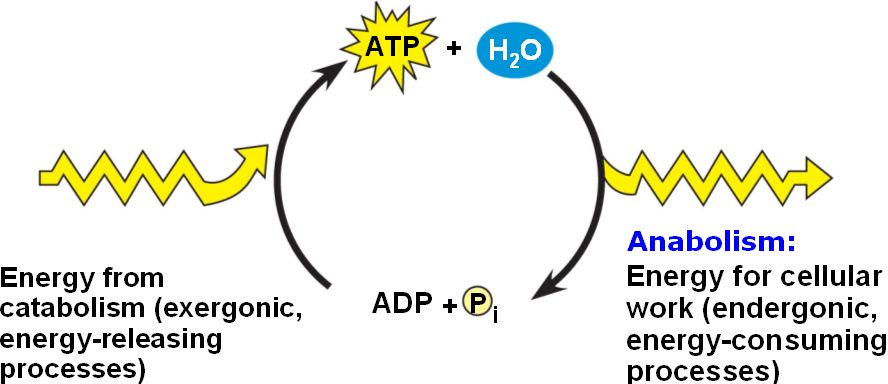
ATP molecules are broken down by hydrolysis (addition of water) to yield inorganic phosphate, adenosine diphosphate (ADP) and energy. The reaction also known as exergonic reaction (release energy). Phosphate bond are one of the main causes in amount of energy released:
|
The major roles of ATP in metabolism are chemical work, transport work and mechanical work:
Chemical work
Chemical work
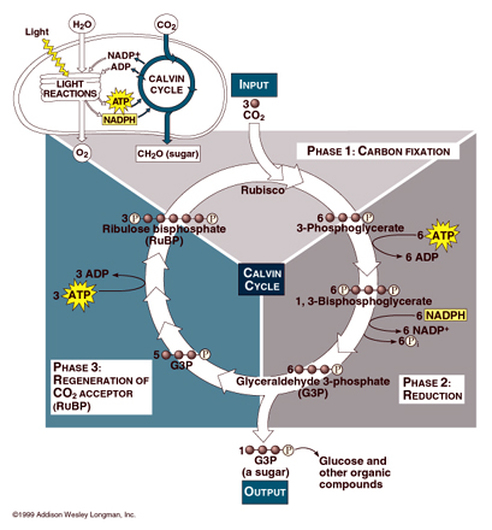
- Synthesis of polymers from monomers by endergonic reaction (consume energy). The energy (ATP) is needed for the reaction to occurs (not spontaneous). For examples in glycolysis (energy-investment phase) and Calvin Cycle (photosynthesis).
Transport work
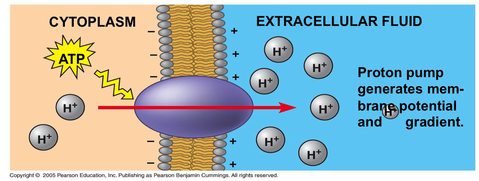
- Active transport either in animal or plant used energy (ATP) to pump the substances across membranes against their concentration gradients.
Mechanical work
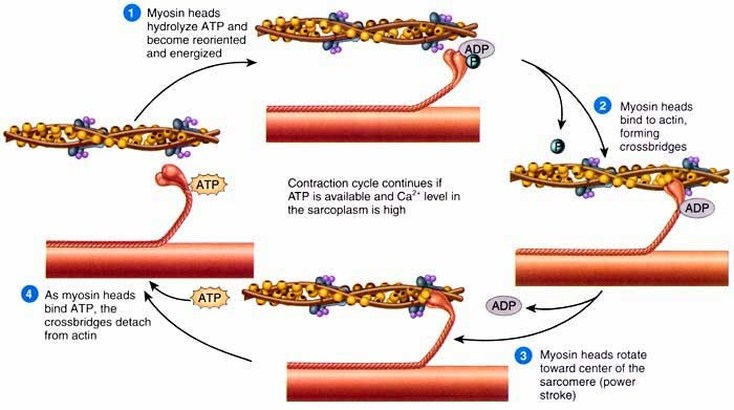
- Supply enough energy (ATP) for muscle contraction such as heart muscle for blood circulation, skeletal muscle for movement and also for beating of cilia and movement of chromosomes during cellular reproduction.
Law of Thermodynamics
|
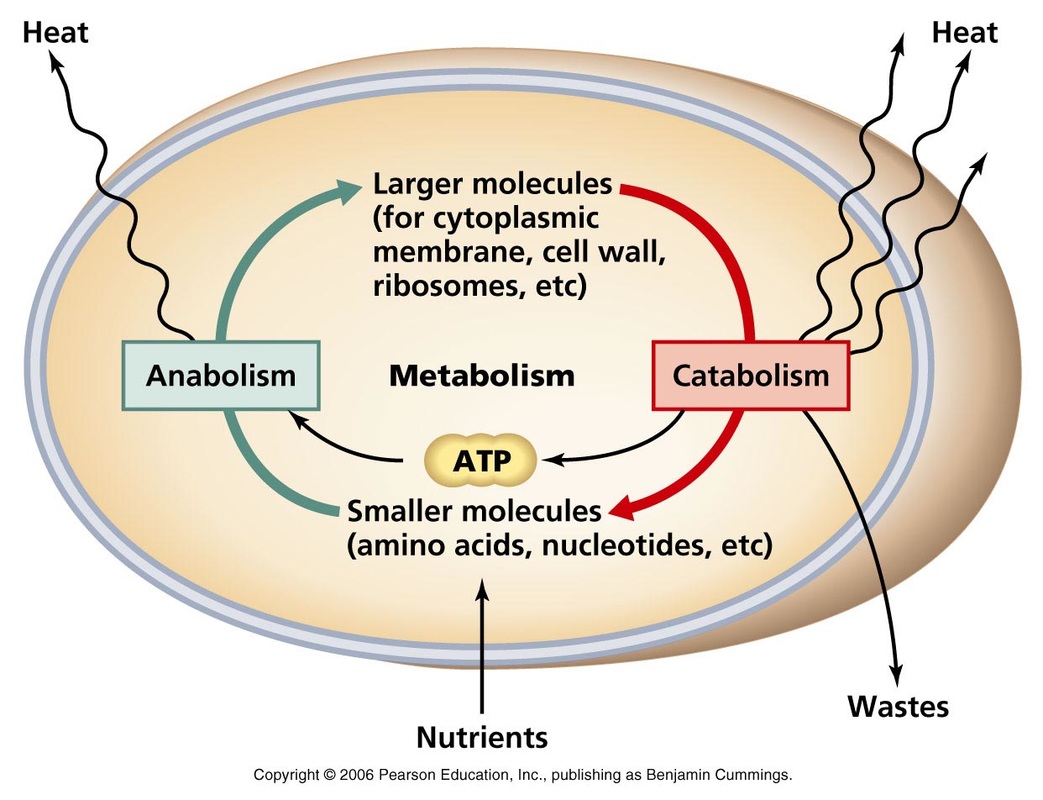
Metabolism
In the Law of Thermodynamics, organism's metabolism is defined as the total of chemical reactions occurs. The chemical reaction of metabolism are organized into metabolic pathway. There are two types of metabolic pathway:
|
Energy
Defined as the capacity to cause change or to do work. Energy can be transferred and transformed but cannot be created and destroyed. There are two kind of energy:
|
Thermodynamics is the study of the energy transformations that occur in a collection of matter. There are two types Law of Thermodynamics:
- The First Law of Thermodynamics: Conservation of energy. Energy can be transferred and transformed, but cannot be created or destroyed.
- The Second Law of Thermodynamics: Every energy transfer and transform increases the entropy of the universe. It is spontaneous reaction causes it occurs without an input of energy.
- Entropy as a measure of disorder or randomness.